MercoPress. South Atlantic News Agency
WHO issues emergency approval for Novavax
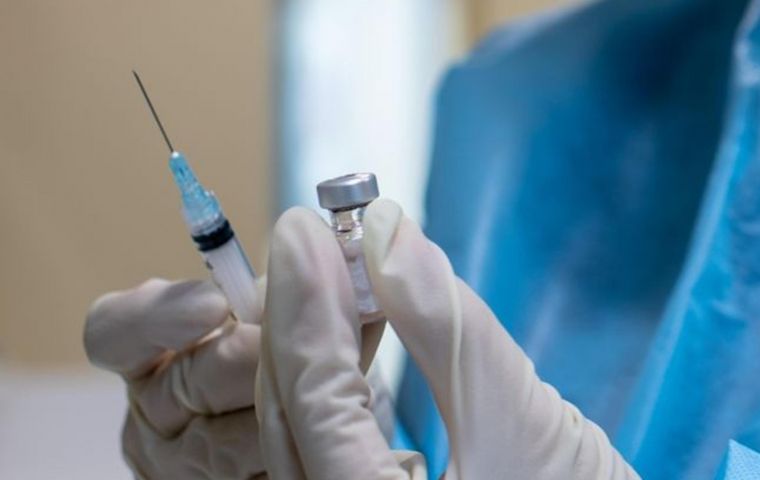 Nuvaxovid has been developed under the same principle used in hepatitis B and whooping cough vaccines.
Nuvaxovid has been developed under the same principle used in hepatitis B and whooping cough vaccines. The World Health Organization (WHO) has issued an emergency approval for the Novavax drug against COVID-19, which has thus become the 10th vaccine available in the fight against the rapidly spreading disease.
The European Medical Agency (EMA) had greenlighted the immunizer, which is traded under the brand Nuvaxovid, earlier this week.
The drug's developers trust this new medication, developed following more conventional methods, will help those still hesitant to getting vaccinated change their minds.
Nuvaxovid, a two-shot treatment to prevent the virus, has been thus added to the WHO's emergency use listing (EUL), it was announced Tuesday. The jab uses a traditional technology involving proteins found on coronavirusspike proteins that trigger an immune response.
Basically, the two-dose vaccine works by introducing a protein that prompts an antibody response, which blocks the ability of future coronavirus to bind to human cells and prevents infection. The protein is combined with Novavax's Matrix-M adjuvant, which is important in enhancing the immune response elicited by the protein antigen.
It is a tried and tested approach, used for decades to vaccinate people against diseases including hepatitis B and whooping cough.
This new vaccine is expected to help overcome vaccine hesitation among people who oppose mRNA drugs out of fears they might change people's DNA or cause infertility. Nuvaxovid also eliminates the argument that it was developed “too quickly.”
WHO's Strategic Advisory Group of Experts on Immunization recommended the new vaccine for use in people over the age of 18, with an interval of three to four weeks between the two doses. “The vaccine should not be administered with an interval of less than three weeks,” it warned. It can be stored at refrigerated temperatures between 2 and 8 degrees Celsius, giving it a logistical advantage in difficult-to-access regions over the mRNA vaccines, which must be stored at ultra-low temperatures.
The Novavax vaccine was shown to be 90.4% effective overall, with 100% efficacy against moderate and severe cases of COVID-19, according to the developers. Pfizer (95%) and Moderna (94%) vaccines and more effective than the one-shot Johnson & Johnson's 66% in clinical trials.
WHO said Nuvaxovid was around 90 percent effective at reducing symptomatic cases of COVID-19in two major clinical studies, one in Britain and the other in the United States and Mexico, involving more than 45,000 people. The company added that the vaccine is 93.2 percent effective against circulating “variants of concern” and “variants of interest.”
Novavax's side effects are relatively mild and similar to commonly reported side effects for Pfizer, Moderna and Johnson & Johnson. The most common side effects were injection site pain and tenderness lasting less than three days, as well as fatigue, headache and muscle pain lasting less than two days.
The WHO said that individuals with a history of anaphylaxis to any component of the vaccine should not take it. The UN health agency also said Novavax was safe for pregnant women or those breastfeeding.




Top Comments
Disclaimer & comment rulesCommenting for this story is now closed.
If you have a Facebook account, become a fan and comment on our Facebook Page!