MercoPress. South Atlantic News Agency
Brazilian gov't releases pediatric COVID-19 vax
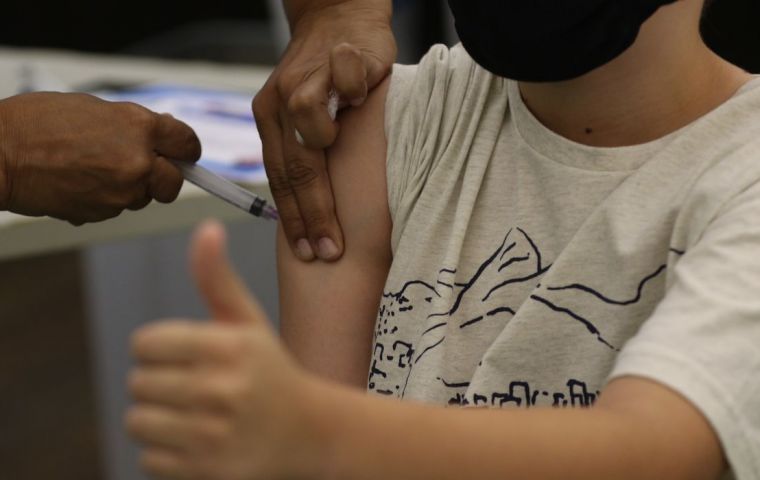 For the time being the vaccine will be available only to patients with comorbidities
For the time being the vaccine will be available only to patients with comorbidities Brazilian health authorities have released Pfizer's pediatric Covid-19 vaccine to be applied to children aged 6 months or older who have comorbidities, it was announced Thursday.
The Health Ministry has released the application of vaccines to children from 6 months to 4 years old who have comorbidities after the Surveillance Agency Anvisa's clearance last month.
In a note dated Oct. 13, the Health Ministry said it will ask the National Commission for Incorporation of Technology in SUS (Conitec) to evaluate the possible expansion of the use of the pediatric vaccine in this age group. Until it is analyzed by the commission, vaccination will be restricted to patients with comorbidities.
The vaccine for children aged 6 months to 4 years old has a different dosage and composition from those used for the previously approved age groups. The vaccine formulation authorized Thursday is to be administered in three 0.2 ml doses (3 micrograms).
The initial two doses should be given 3 weeks apart, followed by a third dose at least 8 weeks after the second dose. The cap of the vaccine bottle will come in wine color, to facilitate identification by vaccination teams and also by fathers, mothers, and caregivers who will take children to be vaccinated.
The use of different lid colors is a strategy to avoid administration errors since the product requires different dosages for different age groups.
“The vaccine has a 12-month shelf life when stored at temperatures between -90°C and -60°C. Once removed from freezing, the unopened vial can be stored in a refrigerator between 2°C and 8°C for a single period of 10 weeks, not exceeding the original expiration date,” Anvisa explained.
(Source: Agencia Brasil)


Top Comments
Disclaimer & comment rulesCommenting for this story is now closed.
If you have a Facebook account, become a fan and comment on our Facebook Page!