MercoPress. South Atlantic News Agency
Tag: vaccine
-
Friday, August 26th 2022 - 17:46 UTC
Brazil greenlights import of vaccine, drug against monkeypox

Brazil's National Health Surveillance Agency (Anvisa) Thursday cleared the Health Ministry in separate but unanimous decisions to import and use the Jynneos / Imvanex vaccine and the drug Tecovirimat, both against monkeypox.
-
Wednesday, December 1st 2021 - 09:22 UTC
“Vaccine”, word of the year for Merriam-Webster: Lookups soar plus a more precise definition

The Merriam-Webster dictionary selected “vaccine” as the word of 2021. The publishing company noted that the word holds particular significance both as a medical term and a vehicle for ideological conflict. Likewise, interest in vaccines started high and grew during the course of 2021.
-
Monday, October 4th 2021 - 18:42 UTC
Arrival into UK made easier: it's all about country of origin being on red list or not

The United Kingdom has launched a simplified system regarding the processing of arriving passengers effective Monday, Oct. 4. Following long queues and no practical improvement, British authorities have decided that arriving travelers are to be measured as coming from a country within the red list... or not.
-
Wednesday, February 24th 2021 - 09:34 UTC
The Falkland Islands receives second batch of Covid-19 vaccines

The Falkland Islands Government has received its second batch of Covid-19 vaccines, supplied by the UK government. These 2,200 doses arrived today via the South Atlantic Airbridge and are of the same Oxford/AstraZeneca type as the first batch received at the beginning of the month.
-
Tuesday, January 12th 2021 - 19:03 UTC
Biden's agenda, Covid-19 and economic aid, cabinet nominations and Trump's impeachment
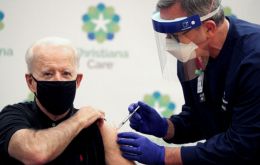
President-elect Joe Biden anticipates three main issues of his starting time in office, cabinet nominations, Trump's impeachment proceedings, and coronavirus and economic aid.
-
Monday, January 4th 2021 - 05:36 UTC
Brazil health regulator approves import of 2 million doses of AstraZeneca vaccine
Brazil's health regulator Anvisa said over the weekend it had approved the import of 2 million doses of the COVID-19 vaccine developed by AstraZeneca and the University of Oxford, although the jab is not yet approved for use in the country.
-
Thursday, December 10th 2020 - 09:10 UTC
UK medicine regulator advises people with allergic reactions to avoid the Pfizer vaccines

Britain's medicine regulator has advised that people with a history of significant allergic reactions do not get Pfizer-BioNTech's Covid-19 vaccine after two people reported adverse effects on the first day of rollout.
-
Wednesday, October 14th 2020 - 09:39 UTC
World Bank approves US$ 12bn for developing countries to purchase Covid-19 vaccines

The World Bank said its executive board approved on Tuesday US$12 billion in new funding for developing countries to finance the purchase and distribution of COVID-19 vaccines, tests, and treatments for their citizens. The financing plan is part of US$160 billion in total resources that the multilateral development lender has pledged to provide to developing countries through June 2021 to help fight the coronavirus pandemic.
-
Friday, October 9th 2020 - 09:45 UTC
EU seals Covid-19 vaccine deal with Johnson & Johnson, securing 1,1 billion doses for its 450 million population

The European Union has sealed a deal with Johnson & Johnson to supply up to 400 million doses of its potential COVID-19 vaccine, as the bloc builds up stocks amid a global scramble to secure shots.
-
Wednesday, September 30th 2020 - 09:45 UTC
World Bank plans US$ 12bn to help poor countries with Covid-19 vaccine purchase and distribution

The World Bank on Tuesday said it has asked its board of directors to approve US$12 billion to help poor countries purchase and distribute eventual vaccines against COVID-19. The bank has already implemented emergency response programs in 111 countries and the extra money, if approved, would be aimed at low- and middle-income countries.
