MercoPress. South Atlantic News Agency
Argentina finds new supplier of vaccines against coronavirus
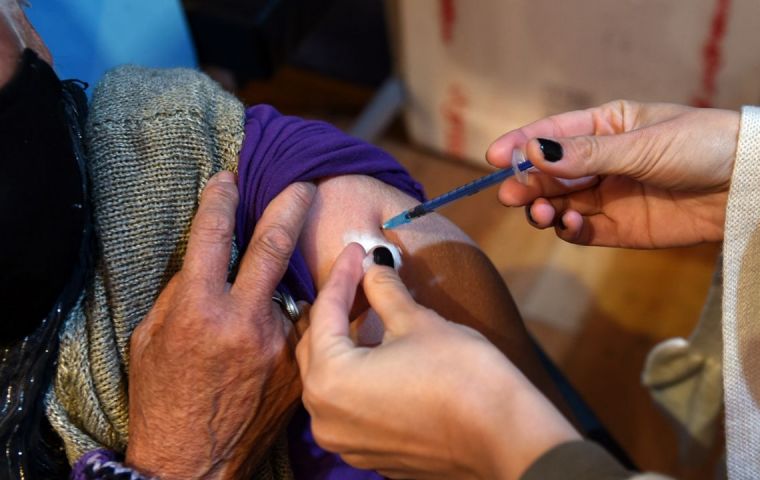 The Cansino vaccine has been “tested in Argentina in recent months,” explained Cafiero
The Cansino vaccine has been “tested in Argentina in recent months,” explained Cafiero Argentine Cabinet Chief Santiago Cafiero Wednesday announced that an agreement with Chinese laboratory Cansino Bio for the supply of aniticoronavirus vaccines had been reached.
“The national government reached an agreement with the Chinese laboratory Cansino Bio for the provision of more vaccines against Covid-19,” Cafiero told the state-run news agency Télam.
Cafiero highlighted the importance of these vaccines which are “in phase 3” of development, in which their efficacy and safety are tested. He also explained that the Chinese laboratory's vaccine has been “tested in Argentina in recent months,” with the participation of Fundación Huiuda.
The Cansino Biologics laboratory has a long history in the development of vaccines and in 2017 it had already developed an immunization against Ebola, a lethal virus that appeared in 1976 and has a high mortality rate.
The Cansino vaccine will facilitate the development of the vaccination campaign since it is applied with a single dose and is stored at a temperature between 2 and 8 degrees. In other words, it does not require freezing, which makes logistics simpler.
This agreement with CansinoBio joins the other five agreements that the Argentine State signed with different laboratories around the world to supply vaccines against the coronavirus.
Between October 2020 and March this year, Argentina has signed contracts with the laboratories that produce the AstraZeneca vaccines, in the United Kingdom; Sputnik V, of the Russian Federation; Sinopharm, from China; Covishield, from India, and the COVAX mechanism carried out through the Gavi Foundation and the World Health Organization (WHO).




Top Comments
Disclaimer & comment rulesCommenting for this story is now closed.
If you have a Facebook account, become a fan and comment on our Facebook Page!