MercoPress. South Atlantic News Agency
Tag: Anvisa
-
Friday, December 17th 2021 - 09:01 UTC
Anvisa wants Brazilian children vaccinated against COVID-19
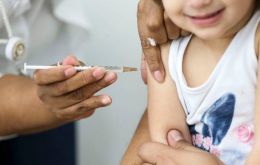
Brazil's National Health Surveillance Agency (Anvisa) Thursday recommended children be vaccinated against COVID-19, despite President Jair Bolsonaro's stance against this type of immunization.
-
Tuesday, December 14th 2021 - 09:49 UTC
Brazil's STF rules proof of vaccination be required to foreign travelers

Brazil's Supreme Federal Court (STF) has ruled over the weekend in favor of a request filed by the opposition to make proof of vaccination against COVID-19 mandatory for all incoming foreign travelers, against the wishes of President Jair Bolsonaro.
-
Thursday, December 2nd 2021 - 09:00 UTC
Travel mate of first Omicron case in Brazil found on Paraguayan border

Sanitary authorities have detected one man who lives in neighboring Foz do Iguaçu and works in Paraguay had boarded the same flight from South Africa Nov. 27 carrying a man infected with the Omicron variant of Covid-19 at a time Brazil's National Health Surveillance Agency (Anvisa) warnings were not yet in force.
-
Wednesday, December 1st 2021 - 09:53 UTC
Brazil reports first two cases of Omicron Covid-19 variant

Brazil's National Health Surveillance Agency (Anvisa) Tuesday reported having detected the first two local cases of the Omicron variant of the coronavirus, which are also the first two infections of this kind in the entire South American continent.
-
Thursday, October 28th 2021 - 09:40 UTC
Over 53% of Brazilians fully vaccinated, report says

Brazil's much-criticized anti-COVID-19 vaccination drive has already reached 53% of the population with full treatment, while 72% of patients have at least taken the initial dose, according to a health report released Wednesday.
-
Monday, June 21st 2021 - 08:53 UTC
Anvisa green-lights sale of 480 tons of medical oxygen to Paraguay

Paraguay's Foreign Ministry Sunday confirmed Brazil's National Health Surveillance Agency (Anvisa) had agreed to allow the sale of 480 tons of oxygen for medical purposes.
-
Tuesday, June 15th 2021 - 09:29 UTC
Anvisa grants shelf-life extension for 3 million doses of Janssen single-dose vaccine in Brazil
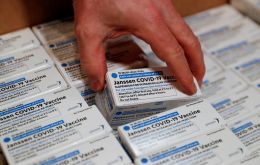
Brazil's National Health Surveillance Agency (Anvisa) on Monday granted a shelf-life extension to 3 million doses of Janssen's Covid-19 vaccine.
-
Thursday, June 10th 2021 - 17:28 UTC
Brazil's Anvisa gives green light to start ButanVac testing pending some paperwork

Brazil's National Health Surveillance Agency of Brazil (Anvisa) Wednesday authorized the Butantan Institute to carry out human tests of the locally-produced ButanVac immunizer.
-
Wednesday, June 9th 2021 - 09:42 UTC
Paraguay needs additional medical oxygen from Brazil

Paraguayan President Mario Abdo Benítez Tuesday reached out to his Brazilian colleague Jair Bolsonaro to negotiate the purchase of medical oxygen above the current quota because it is not enough, according to health authorities.
-
Saturday, June 5th 2021 - 08:40 UTC
Anvisa allows for limited imports of Sputnik V and Covaxin into Brazil

Brazil's National Health Surveillance Agency (Anvisa) Friday approves, albeit with some restrictions, imports of Covaxin and Sputnik V doses for use in up to 1% of the country's population, it was announced.
